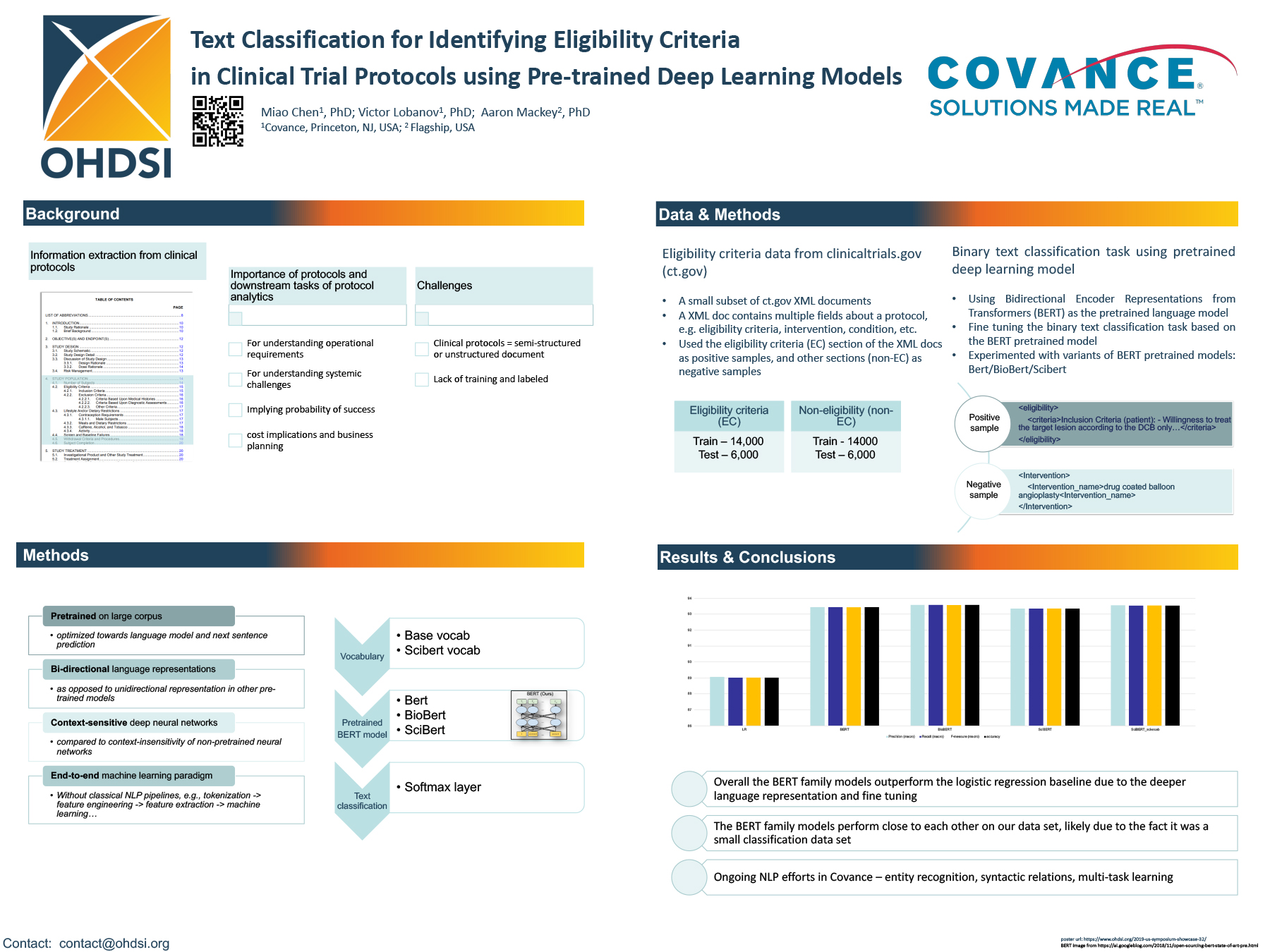
Evaluating the frequency of English language requirements in clinical trial eligibility criteria: A systematic analysis using ClinicalTrials.gov | PLOS Medicine
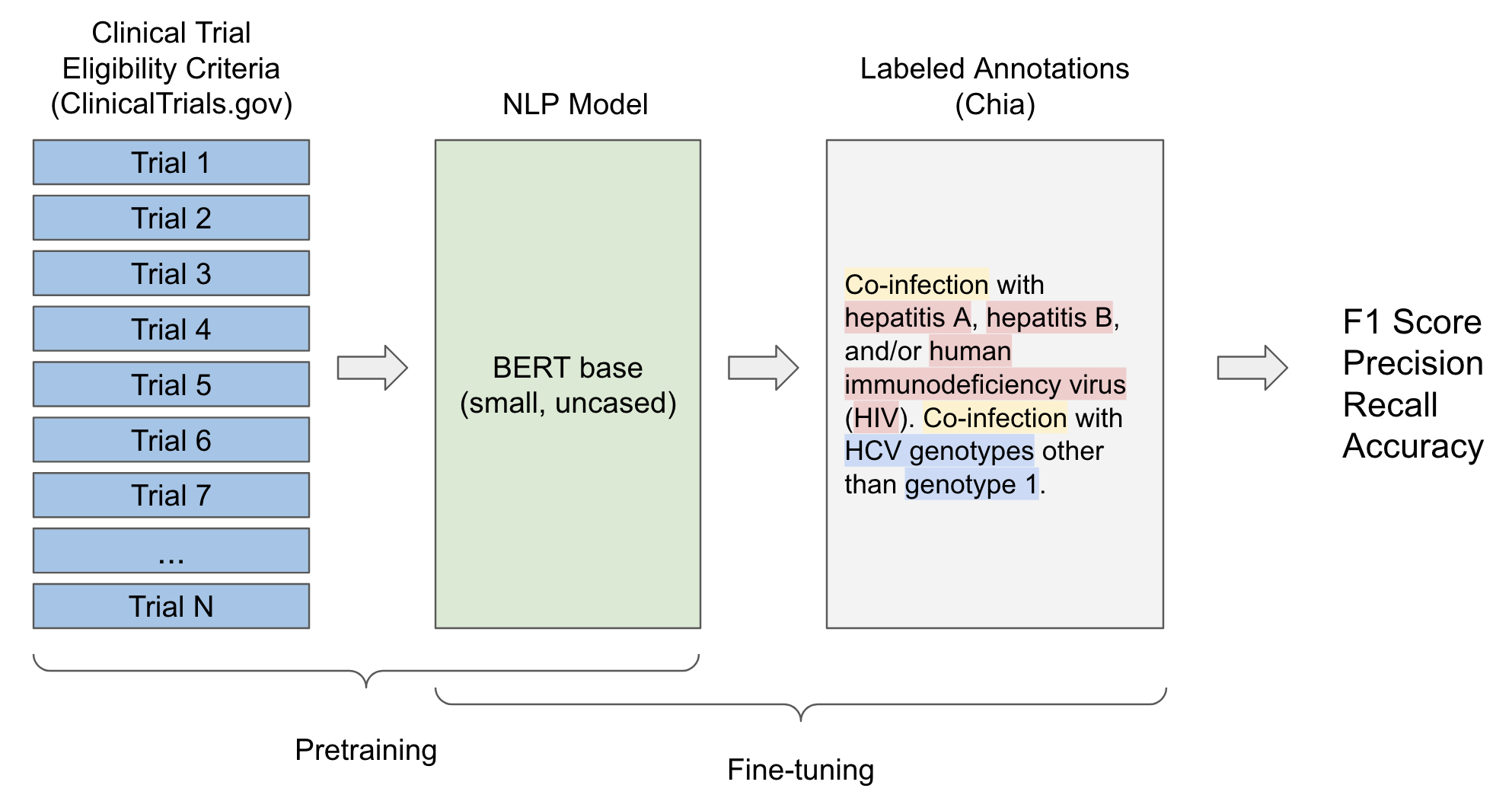
![PDF] Comparison of Eligibility Criteria Between Protocols, Registries, and Publications of Cancer Clinical Trials. | Semantic Scholar PDF] Comparison of Eligibility Criteria Between Protocols, Registries, and Publications of Cancer Clinical Trials. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/62bcc7885dac29517026f3ba577045d3b93dbb7b/2-Table2-1.png)
PDF] Comparison of Eligibility Criteria Between Protocols, Registries, and Publications of Cancer Clinical Trials. | Semantic Scholar

Eligibility Criteria and Representativeness of Randomized Clinical Trials That Include Infants Born Extremely Premature: A Systematic Review - The Journal of Pediatrics

Barriers to Enrollment in Non-small Cell Lung Cancer Therapeutic Clinical Trials - Journal of Thoracic Oncology
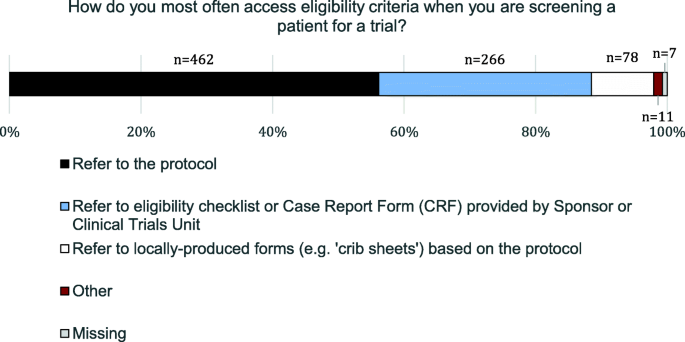
Clinical trial recruiters' experiences working with trial eligibility criteria: results of an exploratory, cross-sectional, online survey in the UK | Trials | Full Text

Analysis of eligibility criteria representation in industry-standard clinical trial protocols - ScienceDirect

Transforming Clinical Trial Eligibility Criteria to Reflect Practical Clinical Application | American Society of Clinical Oncology Educational Book
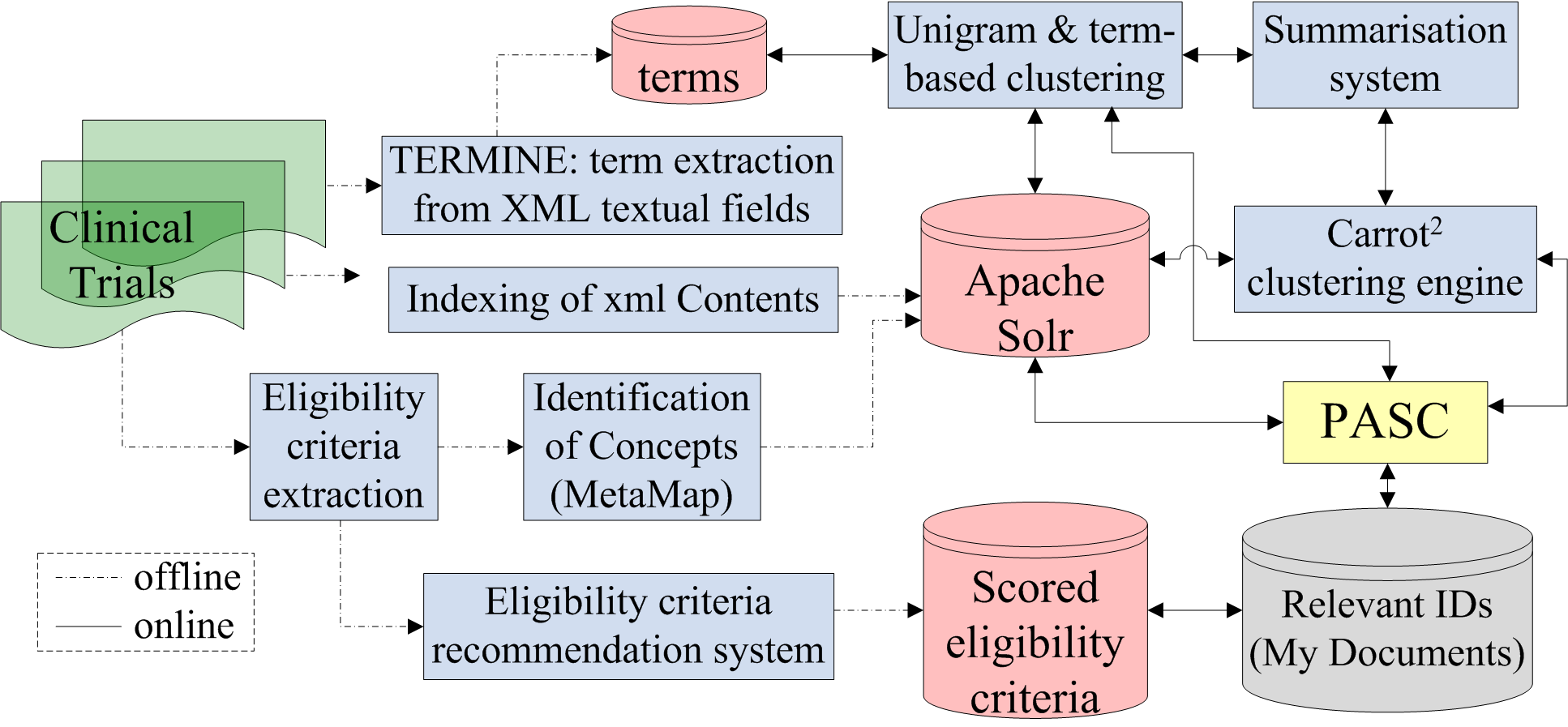
![PDF] Automated classification of eligibility criteria in clinical trials to facilitate patient-trial matching for specific patient populations | Semantic Scholar PDF] Automated classification of eligibility criteria in clinical trials to facilitate patient-trial matching for specific patient populations | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/259f4fca7905dbb313f32a7bcd717bd08785cbd1/4-Figure1-1.png)
PDF] Automated classification of eligibility criteria in clinical trials to facilitate patient-trial matching for specific patient populations | Semantic Scholar

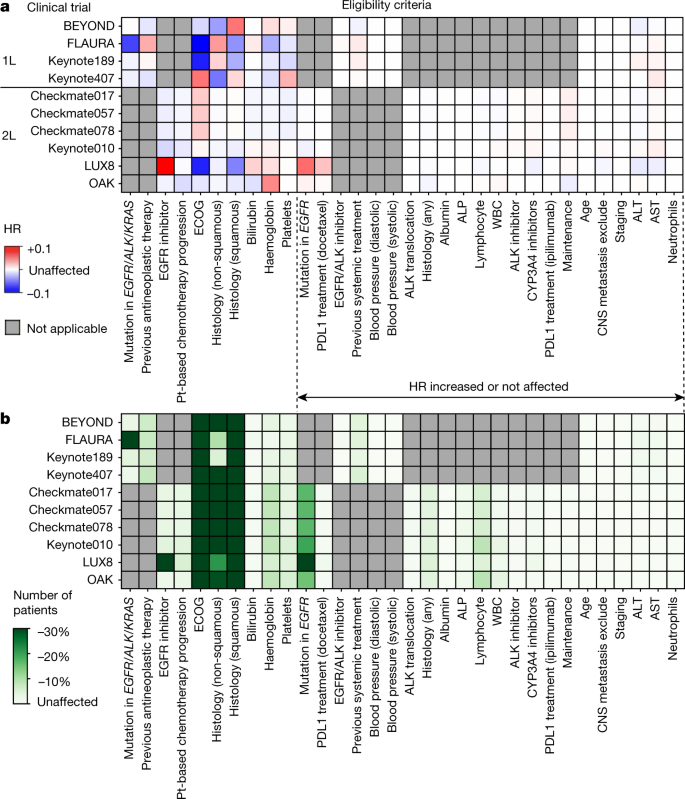
Re-Evaluating Eligibility Criteria for Oncology Clinical Trials: Analysis of Investigational New Drug Applications in 2015 | Journal of Clinical Oncology